Spinal muscular atrophy (SMA) is a hereditary autosomal recessive
disease affecting areas of the nervous system that control voluntary
muscle movement. SMA is identified as a leading genetic disorder causing
mortality among infants. According to Orphanet, one infant in 6,000
births is estimated to suffer from SMA. Furthermore, over 10 million
people in the U.S. are carriers of SMA gene. Depending on the age of
onset and symptoms, spinal muscular atrophy is classified in four main
types. Type 1 SMA is the most severe form, and affects infants aged
between zero and six months. Over 60% of annual SMA cases are of type 1
SMA, wherein infants usually do not survive for more than 2 years. Type 2
SMA is a moderate form affecting children aged between six months and
eighteen months. Patients with Type 3 SMA and Type 4 SMA usually have a
normal lifespan.
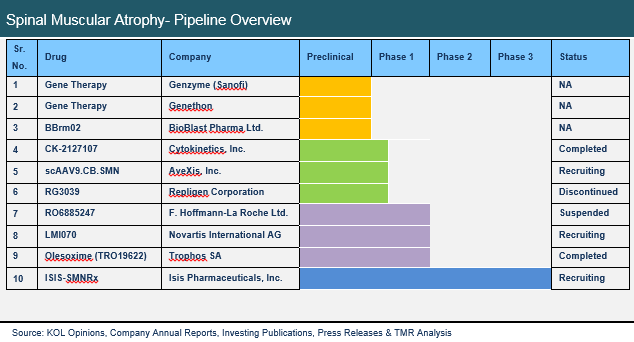
Currently, no cure exists for spinal muscular atrophy, and treatment is directed towards managing common symptoms of SMA. Physiotherapy is strongly recommended in early stages of the disease to extend range of motion and improve muscle flexibility. Other treatment methods are targeted to treat gastrointestinal and respiratory disorders, which are common in SMA patients. The SMA pipeline is dry, with single drug in phase 3, three in phase 2, and one in phase 3 clinical trials. ISIS-SMNRx by Isis Pharmaceutical is an antisense drug that has shown promising results in phase 2 clinical trials. Phase 3 study of ISIS-SMNRx for treatment of Type 1 SMA and Type 2 SMA is expected to complete in 2016. In the United States, the drug is expected to reach annual sales of USD 0.47 billion by 2023. Olesoxime (TRO19622) has completed its phase2/3 trials, and has shown promising results, with potential to effectively treat type 2 and type 3 SMA. Other potential candidates include Olesoxime (TRO19622), LMI070, RO6885247, CK-2127107, scAAV9.CB.SMN and RG3039.
The United States and Europe have higher percentage of diagnosed cases of SMA. Government and private organization in these regions are also involved in creating awareness about the disease. Higher confirmed carrier population would also help in faster market growth in these regions. Furthermore, government incentives for development of therapeutic alternatives for rare diseases also boosts research and development in the SMA landscape.Companies with potential candidates in the spinal muscular atrophy pipeline include AveXis, Inc., Cytokinetics, Inc., F. Hoffmann-La Roche Ltd., Novartis AG, and Isis Pharmaceuticals, Inc.
Spinal Muscular Atrophy Pipeline is segmented as below:
Late Stage Candidates (Phase 3)
ISIS-SMN Rx
Early Stage Candidates (Phase 1 and Phase 2)
Olesoxime (TRO19622)
LMI070
RO6885247
CK-2127107
CB.SMN
RG3039
Request For Sample Report:- http://www.transparencymarketresearch.com/sample/sample.php?flag=S&rep_id=7187

Currently, no cure exists for spinal muscular atrophy, and treatment is directed towards managing common symptoms of SMA. Physiotherapy is strongly recommended in early stages of the disease to extend range of motion and improve muscle flexibility. Other treatment methods are targeted to treat gastrointestinal and respiratory disorders, which are common in SMA patients. The SMA pipeline is dry, with single drug in phase 3, three in phase 2, and one in phase 3 clinical trials. ISIS-SMNRx by Isis Pharmaceutical is an antisense drug that has shown promising results in phase 2 clinical trials. Phase 3 study of ISIS-SMNRx for treatment of Type 1 SMA and Type 2 SMA is expected to complete in 2016. In the United States, the drug is expected to reach annual sales of USD 0.47 billion by 2023. Olesoxime (TRO19622) has completed its phase2/3 trials, and has shown promising results, with potential to effectively treat type 2 and type 3 SMA. Other potential candidates include Olesoxime (TRO19622), LMI070, RO6885247, CK-2127107, scAAV9.CB.SMN and RG3039.
The United States and Europe have higher percentage of diagnosed cases of SMA. Government and private organization in these regions are also involved in creating awareness about the disease. Higher confirmed carrier population would also help in faster market growth in these regions. Furthermore, government incentives for development of therapeutic alternatives for rare diseases also boosts research and development in the SMA landscape.Companies with potential candidates in the spinal muscular atrophy pipeline include AveXis, Inc., Cytokinetics, Inc., F. Hoffmann-La Roche Ltd., Novartis AG, and Isis Pharmaceuticals, Inc.
Spinal Muscular Atrophy Pipeline is segmented as below:
Late Stage Candidates (Phase 3)
ISIS-SMN Rx
Early Stage Candidates (Phase 1 and Phase 2)
Olesoxime (TRO19622)
LMI070
RO6885247
CK-2127107
CB.SMN
RG3039
Request For Sample Report:- http://www.transparencymarketresearch.com/sample/sample.php?flag=S&rep_id=7187

